Fight, Flight, Feel: How the Brain Turns Emotion into Motion
Neuroscience and psychology | Aariana Rao
Emotion as a regulator of motor behaviour
Emotions are traditionally conceptualised as internal experiences or subjective feelings, yet contemporary neuroscience increasingly recognises them as fundamental regulators of motor control. Rather than passively accompanying behaviour, emotional states actively bias which movements are permitted, inhibited, or facilitated. Defensive reactions, such as freezing, fleeing, or fighting, are not merely reflexive responses, but structured motor programmes coordinated by subcortical circuits that have evolved to optimise survival [1].
Central to these processes is the amygdala, a structure located deep within the temporal lobes that integrates sensory information related to threats and communicates with downstream regions, including the periaqueductal grey, to initiate context-specific motor patterns. This circuitry enables defensive actions to be executed rapidly, often before conscious awareness emerges, illustrating that motor preparation and action frequently precede the subjective experience of emotion [2].
Positive emotional states engage overlapping neural circuits but exert contrasting functional effects. Signals associated with safety, reward, or curiosity reduce inhibitory tone within motor circuits. In this way, emotions can be understood as dynamic modulators of the action repertoire, shaping both the likelihood and scope of motor outputs in response to internal states and environmental demands [3].
Fear, stress, and avoidance exemplify emotional conditions that restrict behavioural options, biasing action towards defensive or habitual motor programmes. In contrast, positive emotions broaden the range of available actions by loosening inhibitory constraints within motor networks. Together, these findings underscore that emotion functions not only as an internal experience, but as a core biological mechanism that regulates behavioural potential by shaping the selection, probability, and execution of action [2], [3].
The neurobiology of emotional action control
Emotional processing has traditionally been described as a linear sequence in which an individual experiences an emotion, cognitively appraises it, and then acts. While intuitive, this framework oversimplifies the underlying biology. A more accurate account is that emotions bias the body’s readiness for action, with the brain preparing, inhibiting, or facilitating movement in accordance with emotional state [3], [4]. Although the link between emotion and motor systems is well established, it is often overlooked in everyday interpretations of emotional experience, potentially reinforcing behavioural cycles that are difficult to disrupt.
Emotions are not grounded solely in bodily sensations, but arise through the integration of internal states with perceptual representations of external objects and events. This multimodal integration underpins the intentionality of emotion and shapes the actions it elicits. To put it simply, if actions are considered a door, emotion functions as the key that determines whether, and how, that door can be opened [3], [4].
A core principle underlying this framework is that action requires a release from inhibition. Intentional inhibition refers to the voluntary suppression of an action despite a strong motivation to act. A common example is drafting an email and hesitating before pressing ‘send’: the intention and preparatory motor processes are already engaged, yet the action is deliberately withheld [5]. Crucially, this form of inhibition is internally generated rather than triggered by external cues, distinguishing it from classic paradigms that rely on externally imposed stop signals. While intentional inhibition prevents motor execution, it does not abolish the underlying motivation. For example, refraining from acting out of anger does not eliminate the desire to express it. This process is typically associated with three experimental components: (1) an urge to act, (2) the simultaneous recognition of a reason to resist that urge, and (3) frustration arising from the failure to execute the intended action [5].
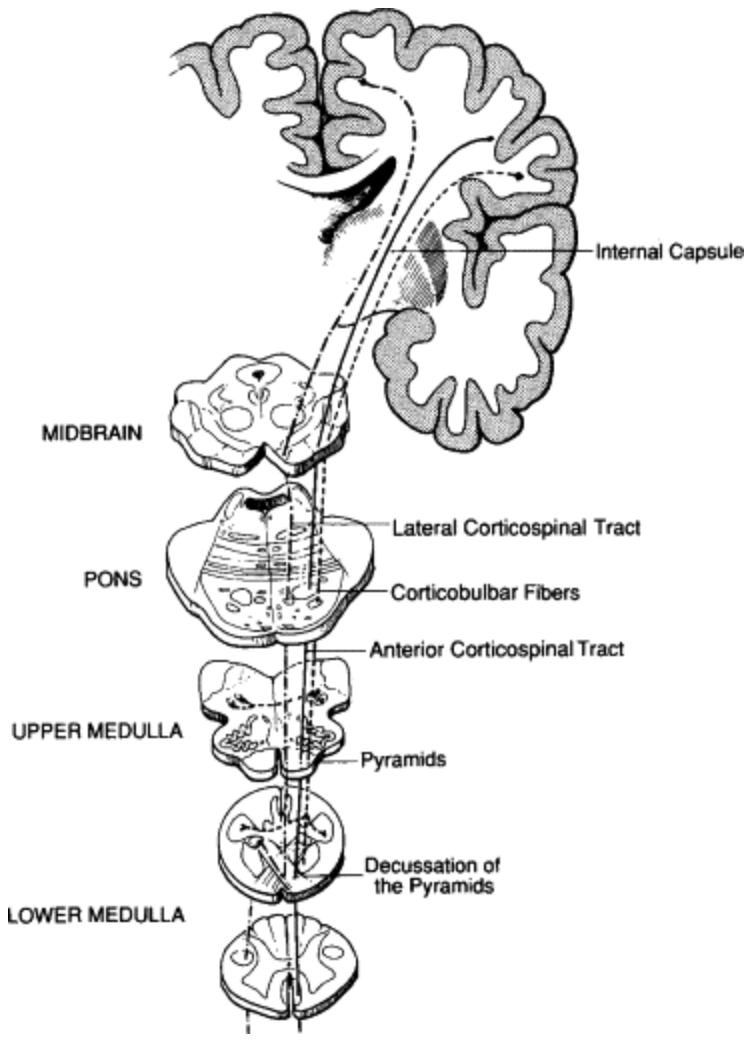
Figure 1: An overview of the corticospinal tract, including corticospinal and corticobulbar fibres [6].
Glossary
Anterior: a position toward the front of the body or brain relative to other structures.
Aversive: stimuli or emotional states that are unpleasant and promote avoidance, withdrawal, or defensive behavioural responses.
Basolateral: a region located toward the base and side of a structure, most commonly used to describe a subdivision of the amygdala involved in sensory integration and emotional learning.
Decussate: the crossing of nerve fibres from one side of the central nervous system to the opposite side, typically at a defined anatomical junction.
Dorsal: a position toward the back or upper surface of the body or brain, depending on anatomical context.
Dorsolateral: a location toward the upper (dorsal) and lateral regions of the brain, frequently implicated in executive control and cognitive regulation.
Downstream: refers to neural processes, effects, or regions that occur later in a signalling pathway, following earlier (upstream) neural activity.
Ipsilaterally: refers to structures or processes occurring on, or projecting to, the same side of the body or brain.
Lateral: a location or orientation away from the midline and toward the sides of the body or brain.
Locomotor: pertaining to the control or execution of whole-body movement, particularly movement through space.
Nociceptive: the detection, transmission, and processing of signals arising from potentially damaging or painful stimuli.
Orofacial: relating to the muscles and neural control of the mouth and face, including behaviours such as chewing, vocalisation, and facial expression.
Periaqueductal grey: a midbrain structure surrounding the cerebral aqueduct that integrates emotional, autonomic, and nociceptive information to coordinate defensive behaviours and modulate pain.
Tonic immobility: a defensive state characterised by involuntary motor inhibition and profound stillness (total unresponsiveness), typically occurring in response to inescapable threat.
Ventromedial: a position toward the lower (ventral) and central (medial) aspects of the brain, often associated with emotion regulation and decision-making functions.
The corticospinal tract, also known as the pyramidal tract, is the principal neural pathway mediating voluntary motor control. It connects the cerebral cortex to the spinal cord via the brainstem, enabling coordinated movement across skeletal muscles, as outlined in Figure 1. Originating in the frontoparietal cortices, corticospinal fibres converge into compact bundles as they descend through the brainstem, becoming increasingly condensed at the level of the pons [7]. Upon reaching the medulla, approximately 70–95% of fibres decussate at the pyramidal decussation, crossing to the contralateral side of the nervous system. The remaining fibres form the anterior corticospinal tract, which descends ipsilaterally and typically terminates at the lower thoracic levels of the spinal cord, located in the mid-back region. Within the spinal cord, fibres travel through the anterior and lateral corticospinal tracts before synapsing onto anterior horn neurons of the grey matter. These neurons project to axial and limbal muscles, ultimately enabling voluntary motor function [7].
The basal ganglia are a group of subcortical nuclei situated deep within the forebrain that play a central role in the regulation of motor control. Beyond movement, they contribute to reasoning, adaptive behaviour, reward-based learning, motivation, and cognitive processes involved in initiating and completing actions. Their close relationship with the cerebral cortex allows them to be organised into discrete, functionally specialised cortico-basal ganglia loops [8]. Rather than providing diffuse cortical access to the motor system, these circuits support parallel processing across motor and non-motor domains. Consequently, damage to basal ganglia circuits linked to motor cortical regions primarily produces motor deficits, whereas disruption of non-motor circuits results in higher-order cognitive and behavioural impairments [8].
The interaction between emotional and motor systems is therefore mediated by specialised neural circuits that integrate sensory, cognitive, and affective information to guide behaviour [1]. A key node in this interface is the nucleus accumbens, a structure within the basal ganglia that is well positioned to translate emotional states into action. The nucleus accumbens receives convergent input from emotion-related regions, including the amygdala, and projects to non-motor related structures, enabling emotional signals to influence locomotor and orofacial motor responses [1].
Fear as a survival motor programme
Fear is widely regarded as a primary emotion, with its neural underpinnings classically linked to the amygdala. It is defined as an aversive emotional state arising from the anticipation or perception of actual or potential danger. One of the most extensively studied models of emotional learning is classical Pavlovian fear conditioning, particularly in animal research [9]. In this paradigm, a neutral stimulus, commonly a tone, is repeatedly paired with an aversive unconditioned stimulus, such as a mild electric shock. Following conditioning, the previously neutral stimulus alone elicits species-specific fear responses, including freezing or escape behaviours, autonomic changes such as increased heart rate and blood pressure, and neuroendocrine responses such as stress hormone release. Through this process, newly learned threats gain access to evolutionarily conserved neural programmes that support adaptive responses to danger [9].
Extensive evidence demonstrates that lesions of the amygdala disrupt both the acquisition and expression of conditioned fear. Within the amygdala, the basolateral complex serves as a critical site of sensory convergence and stimulus association, integrating cortical and subcortical inputs during fear learning [9]. In contrast, the central nucleus projects to downstream regions involved in executing fear responses, including the hypothalamus, and is widely considered a primary output pathway for conditioned fear. Lesions to either region disrupt both learning and expression of fear responses, supporting this functional organisation [9].
Fear-related behaviours are further mediated by projections within the prefrontal cortex–amygdala–periaqueductal grey pathway, which exerts top-down modulation over the descending inhibitory pain system. The prefrontal cortex processes nociceptive and aversive information and maintains reciprocal connections with the amygdala, while the periaqueductal grey integrates emotional, cognitive, and behavioural responses to threat [10]. Studies of fear-conditioned analgesia, which is the reduction in pain sensitivity upon re-exposure to a conditioned aversive stimulus, have provided key insights into endogenous pain suppression and its relationship with fear [10]. The dorsal periaqueductal grey plays a central role in this process, mediating both conditioned and unconditioned fear responses through activation of descending inhibitory pathways. The basolateral and central nuclei of the amygdala provide major output to the dorsal periaqueductal grey, linking emotional processing with pain modulation. Electrophysiological evidence further indicates that potentiation of the amygdala–periaqueductal grey pathway is associated with anxiety-like behaviours in animal models [10].
The phrase “fight or flight” is commonly used to describe behavioural responses to threat, yet it is often misinterpreted as reflecting conscious emotional choice. In reality, these responses represent the activation of distinct, evolutionarily conserved motor programmes. Humans possess a repertoire of innate defensive behaviours, collectively termed the defence cascade, that are automatically recruited in response to threat [11]. Fight and flight represent active defensive strategies, freezing reflects a temporary suspension of action, and tonic immobility emerges as a last-resort response to inescapable danger. Each state is characterised by a distinct neural signature involving coordinated activity across the amygdala, hypothalamus, periaqueductal grey, and autonomic nuclei [11].
While many animals rapidly return to baseline functioning once danger has passed, humans often remain locked into recurrent defensive patterns long after the original threat or trauma has resolved. Understanding the neural components that generate these responses is therefore critical for developing effective therapeutic interventions [11]. Such approaches aim to selectively modulate elements of defensive circuitry, shifting the associated neural pattern and corresponding mind-body state. Central to this framework is the recognition that subjective fear emerges after the body has already been biased toward action, constraining the range of available behaviours before conscious emotional awareness fully develops [11].
Stress-induced biasing of action control and decision-making
Stress is a pervasive feature of everyday life and can be conceptualised as a state of sustained action readiness without resolution. Like fear, stress engages evolutionarily conserved defensive systems that bias the body toward action before conscious deliberation occurs. Stressful situations vary in intensity, producing a spectrum of emotional and behavioural responses closely aligned with the defence cascade [12]. Individuals are frequently required to make decisions under stress, particularly when perceived threat is high or when time and cognitive resources are limited. Under these conditions, decision-making strategies often shift, resulting in choices that differ markedly from those made in low-stress contexts. These effects are best understood through the lens of motor biasing rather than impaired reasoning alone [12].
From an evolutionary perspective, the stress response enhances survival in situations requiring rapid action. Physiological adaptations, including increased heart rate, deeper respiration, elevated glucose availability, thermoregulatory sweating, and heightened muscle tension, prepare the body for movement and prioritise speed over deliberation [12]. At the cognitive level, stress biases behaviour toward intuitive, low-effort strategies. Under normal conditions, intuitive impulses are evaluated by prefrontal control systems that allow for flexible planning and inhibition. Under stress, however, top-down control is weakened, allowing default and habitual motor programmes to be released with minimal modification [12].
Neurobiologically, stress impairs prefrontal cortex functions such as working memory, attention regulation, and response inhibition, while increasing reliance on bottom-up signalling from subcortical regions including the amygdala and basal ganglia. Neuroimaging studies show that acute stress reduces connectivity between ventromedial and dorsolateral prefrontal cortex regions involved in self-control, limiting goal-directed action selection and favouring habitual responses [12].
Critically, the behavioural effects of stress are best characterised as a narrowing of available actions rather than a loss of rationality. Stress constrains behavioural freedom by biasing the motor system toward a restricted set of survival-relevant responses before conscious awareness emerges. Although this may be subjectively experienced as feeling ‘trapped’, it reflects a body that has already committed to defensive pathways in advance of conscious choice [12].
Neural effects of positive emotions on behavioural flexibility
Research has historically prioritised threat-related emotions and their effects on behaviour, yet emotion-action coupling cannot be fully understood without considering positive emotional states. In contrast to fear and stress, which narrow behavioural options, positive emotions broaden the momentary thought–action repertoire, supporting the accumulation of enduring personal resources across physical, cognitive, social, and psychological domains [13].
Positive emotions bias the motor system toward approach and continued engagement rather than defensive withdrawal. Theoretical frameworks consistently suggest that positive affect promotes exploration, persistence, and interaction with the environment. Whereas neutral states may leave individuals disengaged, positive emotions increase the likelihood of approaching novel objects, people, or situations, supporting adaptive development at both individual and species levels [13].
Positive emotional states generate internal signals that promote initiation or continuation of action, although these signals are less rigidly specified than those associated with threat. Sensory pleasure motivates approach toward biologically relevant stimuli, while diffuse positive moods encourage persistence in ongoing activities [13]. Discrete emotion theories link joy with aimless activation, interest with attentional engagement, and contentment with behavioural quiescence. Although these tendencies are less specific, they nonetheless reflect consistent links between emotional state and motor expression, as seen in energetic or animated behaviours associated with excitement and joy [13].
Positive emotions typically arise in non-life-threatening contexts, where rapid behavioural narrowing is unnecessary. Rather than suppressing alternative actions, positive affect expands behavioural possibilities, increasing flexibility and choice. Emotions such as joy, interest, contentment, excitement, pride, and love thus widen the set of available actions, directly counteracting the constraining effects of fear and stress [13].
While threat-related emotions confer immediate adaptive benefits, the broadened repertoires associated with positive emotions yield indirect and long-term advantages. Through exploration and engagement, positive emotional states build durable personal resources that persist beyond the transient emotional experience. These resources form reserves that can later be drawn upon when individuals encounter stress or threat, enhancing resilience and adaptive capacity over time [13].
Conclusion
Across emotional states ranging from fear and stress to joy and excitement, a unifying principle emerges: emotions do not simply follow action, but actively shape the body’s readiness to act. Rather than functioning as conscious directives, emotions bias motor systems by preparing, constraining, or releasing specific patterns of behaviour through coordinated neural circuits involving the prefrontal cortex, basal ganglia, amygdala, periaqueductal grey, and corticospinal pathways. Threat-related emotions engage evolutionarily conserved defence programmes that narrow behavioural options to prioritise survival, whereas positive emotions expand the action repertoire, fostering exploration, learning, and resilience. Stress represents a prolonged state of action readiness in which inhibitory control is weakened and behavioural freedom becomes constrained before conscious awareness emerges. Together, these findings challenge the traditional view of emotion as a purely subjective experience that precedes action. Instead, emotion emerges as a dynamic biological mechanism that sculpts the action landscape itself, determining not only what a person feels, but what they are able to do.
[1] A. Braine and F. Georges, “Emotion in action: When emotions meet motor circuits,” Neurosci. Biobehav. Rev., vol. 155, p. 105475, Dec. 2023, doi: 10.1016/j.neubiorev.2023.105475.
[2] G. Šimić et al., “Understanding Emotions: Origins and Roles of the Amygdala,” Biomolecules, vol. 11, no. 6, p. 823, May 2021, doi: 10.3390/biom11060823.
[3] B. L. Fredrickson and C. Branigan, “Positive emotions broaden the scope of attention and thought-action repertoires,” Cogn. Emot., vol. 19, no. 3, pp. 313–332, May 2005, doi: 10.1080/02699930441000238.
[4] A. B. Eder, “A perceptual control theory of emotional action,” Cogn. Emot., vol. 37, no. 7, pp. 1167–1184, Nov./Dec. 2023, doi: 10.1080/02699931.2023.2265234.
[5] E. Filevich, S. Kühn, and P. Haggard, “Intentional inhibition in human action: the power of ‘no,’” Neurosci. Biobehav. Rev., vol. 36, no. 4, pp. 1107–1118, Apr. 2012, doi: 10.1016/j.neubiorev.2012.01.006.
[6] R. J. Love and W. G. Webb, Neurology for the Speech-Language Pathologist, 2nd ed. Oxford, England: Butterworth-Heinemann, 2013.
[7] A. L. Natali, V. Reddy, and B. Bordoni, “Neuroanatomy, corticospinal cord tract,” in StatPearls, Treasure Island, FL, USA: StatPearls Publishing, 2025.
[8] G. Leisman, O. Braun-Benjamin, and R. Melillo, “Cognitive-motor interactions of the basal ganglia in development,” Front. Syst. Neurosci., vol. 8, p. 16, Feb. 2014, doi: 10.3389/fnsys.2014.00016.
[9] G. M. Goodwin and R. Norbury, “The Amygdala and Fear,” in Stress: Concepts, Cognition, Emotion, and Behavior, G. Fink, Ed., San Diego, CA, USA: Academic Press, 2016, pp. 305–310.
[10] R. K. Butler, L. Nilsson-Todd, C. Cleren, I. Léna, R. Garcia, and D. P. Finn, “Molecular and electrophysiological changes in the prefrontal cortex-amygdala-dorsal periaqueductal grey pathway during persistent pain state and fear-conditioned analgesia,” Physiol. Behav., vol. 104, no. 5, pp. 1075–1081, June 2011, doi: 10.1016/j.physbeh.2011.05.028.
[11] K. Kozlowska, P. Walker, L. McLean, and P. Carrive, “Fear and the Defense Cascade: Clinical Implications and Management,” Harv. Rev. Psychiatry, vol. 23, no. 4, pp. 263–287, July/Aug. 2015, doi: 10.1097/HRP.0000000000000065.
[12] R. Yu, “Stress potentiates decision biases: A stress induced deliberation-to-intuition (SIDI) model,” Neurobiol. Stress, vol. 3, pp. 83–95, Feb. 2016, doi: 10.1016/j.ynstr.2015.12.006.
[13] B. L. Fredrickson, “The Role of Positive Emotions in Positive Psychology,” Am. Psychol., vol. 56, no. 3, pp. 218–226, Mar. 2001, 10.1037//0003-066x.56.3.218.

Aariana is completing her Masters in Biomedical Science. She’s passionate about neuroscience, neuropsychology, and exploring how brain biology shapes human behaviour. Outside of university, she can usually be found curled up with a good book.
